Malin Ardhammar
Department of Physical Chemistry
CHALMERS UNIVERSITY OF TECHNOLOGY
Göteborg, Sweden
Ph.D thesis title:
"Membrane Orientation Studies with Polarized Light Microscopy"

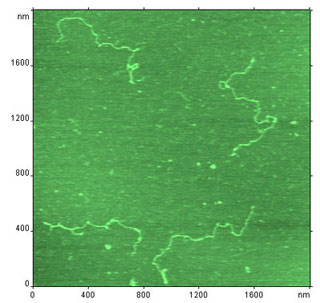
AFM image of the complex of DNA with D,D-[Ru2(phen) 4BD]Cl4 (1:20 bp) immobilized on NiCl2-pretreated mica.
__________________________________________
The work was supported by the Royal Swedish Academy of Sciences through
funds from the research exchange and collaboration programme with Russia.
During her visit of to the SPM joint research group (22.02.99-12.03.99) Malin Ardhammar studied intercalation of D,D-[Ru2(phen)4BD]Cl4* into DNA plasmid FX-174 by means of atomic force microscopy.
Drugs interact with DNA in a multitude of ways. The elucidation of the nature of interaction, for instance if the drug orients along one of the grooves of DNA or intercalates between the base pairs, is of vital importance in the development of new DNA-directed drugs. D,D-[Ru2(phen)4BD]Cl4 is a potential bis-intercalator developed at the Department of Physical Chemistry, Chalmers University of Technology. This ruthenium complex has been shown to possess highly interesting features both in the interaction with DNA and with some rapidly dividing cell lines. Its interaction with DNA has been examined mainly with spectroscopic tools, such as absorption, fluorescence, linear and circular dichroism. The results indicate indeed the mode of binding to be bis-intercalative. However, since steric and electrostatic interactions might be considered to disfavour bis-intercalation for this compound, there is a need for further evidence for the binding mode. With intercalative agents present, the DNA extends, whereas there is no extension with only groove binding agents present. The goal of the research was to measure an elongation (or to verify the absence of elongation).
DNA was immobilized on flat mica surface and imaged in AFM, which allowed length measurements. Experiments were carried out for DNA plasmid FX-174 with a length of 5386 basepairs (bp) and its complexes with ethidium bromide (1:10 bp) and D,D-[Ru2(phen)4BD]Cl4 (1:10 bp and 1:20 bp).
Using only the difference between the two experiments with different ruthenium:DNA molar ratio, the DNA is elongated by 2.45 Å for every intercalator. Ethidium, on the other hand, elongated the DNA by only 0.9 Å for every intercalator.
______________________________________
* D,D-[Ru2(phen)4BD]Cl4 (Mr: 1740), where the bridging ligand BD is N,N'-bis(12-cyano-12,13-dihydro-11H-cyclopentadipyrido[b:3,2-h:2',3'-j]phenazine-12-carbonyl)-1,4-diaminobutane
and phen = 1,10-phenanthroline, is a dimeric analogue to the well-known
luminescent DNA probe D-[Ru(phen)2dppz]4+.